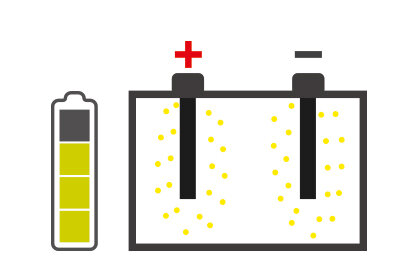
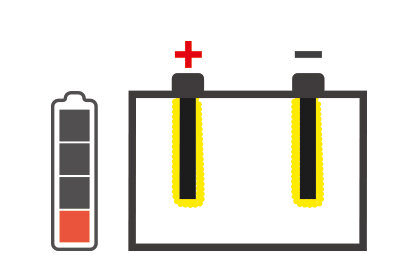
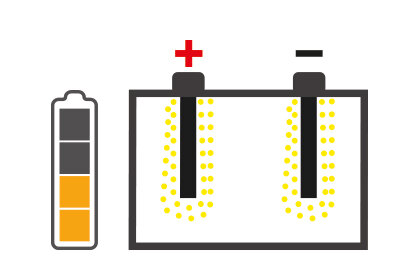
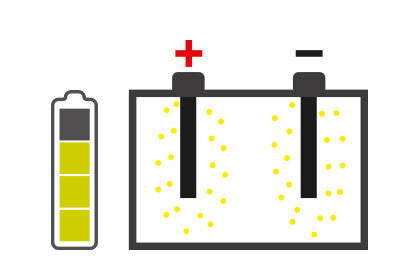
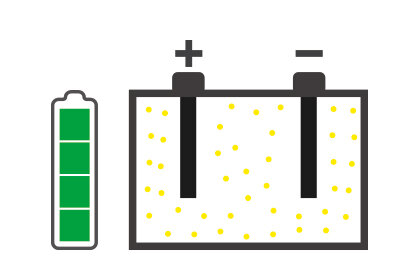
It is essential to know about those situations that can hinder battery performance: one of these is sulfation, which can degenerate battery performance until it is no longer usable.
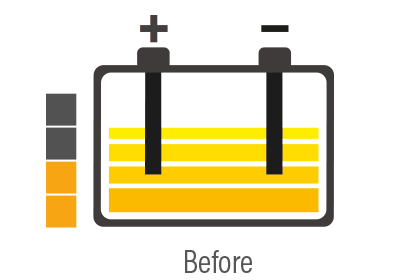
Battery sulfation is caused by many different facts, but the battery remaining for long periods without being used, and therefore subjected to self-discharging, is probably the most common. In addition, the continually growing number of devices in today's vehicles that constantly require energy even when the engine is switched off, for example alarm systems, can cause the battery to discharge progressively and create the conditions for sulfation.
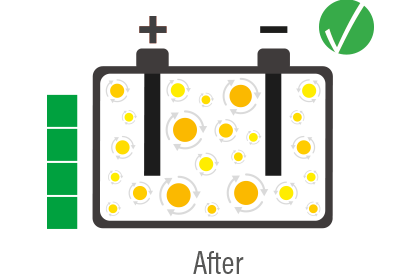
The concept of desulfation follows as a consequence of this, i.e. a process that allows to restore the functionality of the battery by increasing its life and eliminating the necessity of replacing it beforehand.